Two Targets, One Pathway: The Metabolomic Logic Behind MAT2A/PRMT5 Combination Therapies
The next breakthrough in cancer treatment may come from attacking two targets within the same metabolic pathway. MAT2A and PRMT5 inhibitor combinations represent a new paradigm in precision oncology, using metabolic dependencies to create synthetic lethal interactions that spare normal cells while eliminating cancer cells. With major pharmaceutical partnerships validating this approach, understanding the metabolomic rationale behind these combinations is crucial for successful development.
The Metabolic Foundation
The scientific elegance of MAT2A/PRMT5 combinations lies in their complementary mechanisms within the methionine cycle. MAT2A (methionine adenosyltransferase 2A) catalyzes the production of S-adenosylmethionine (SAM), the universal methyl donor required for PRMT5 activity. Meanwhile, accumulated methylthioadenosine (MTA) in MTAP-deleted cancers directly inhibits PRMT5 by competing with SAM for enzyme binding.
This creates a metabolic pincer movement: MAT2A inhibition depletes the SAM needed for PRMT5 function, while MTA accumulation simultaneously blocks remaining PRMT5 activity. Normal cells with intact MTAP can metabolize MTA and maintain SAM levels, making them relatively resistant to this dual inhibition. Cancer cells with MTAP deletions become trapped in a metabolic vise that leads to cell death.
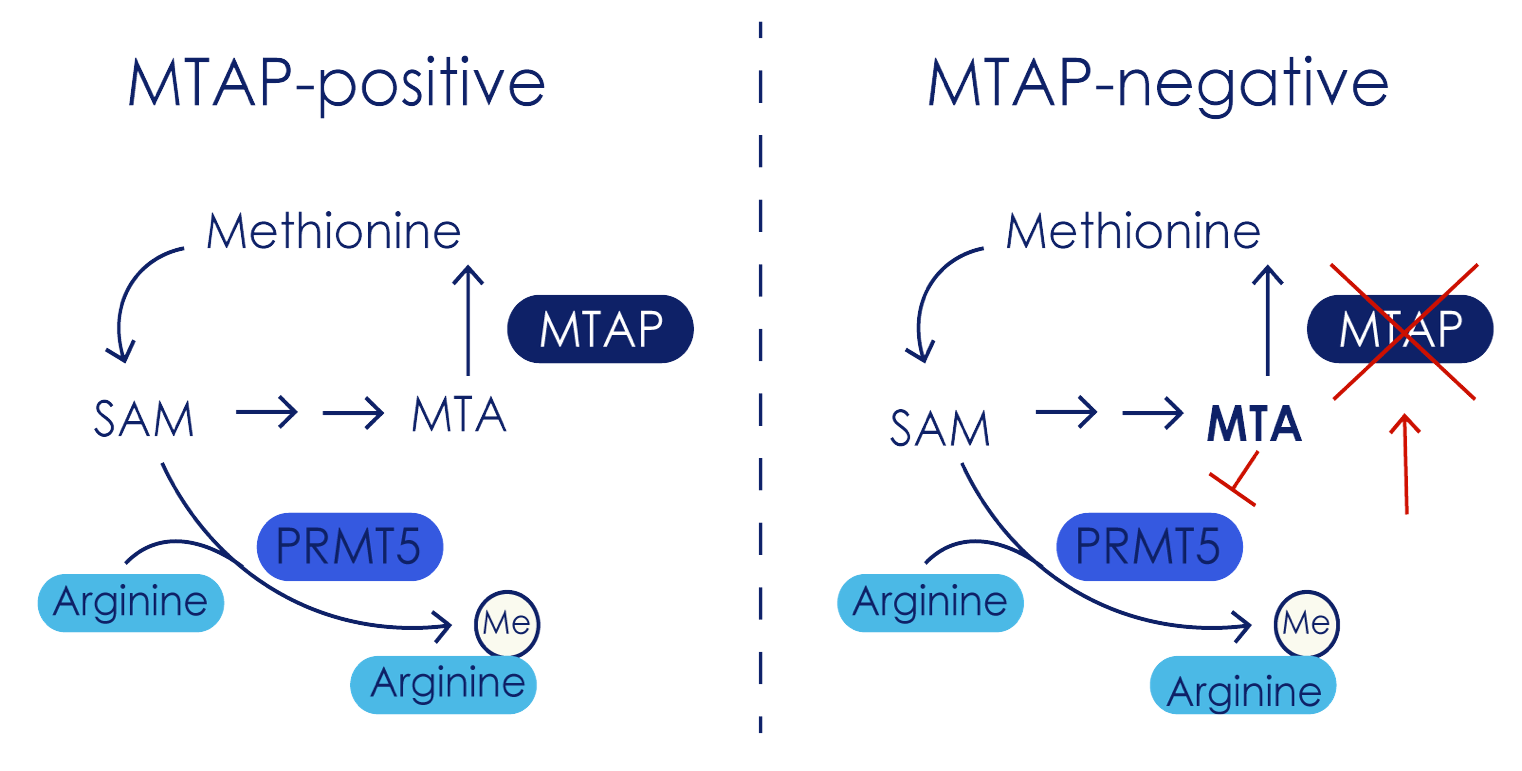
The quantitative relationship is particularly compelling. PRMT5 exhibits 40-fold weaker affinity for SAM compared to MTA, making it exquisitely sensitive to the SAM/MTA ratio. Even modest changes in this ratio, achievable through MAT2A inhibition, can dramatically shift the metabolic balance toward PRMT5 inhibition in vulnerable cancer cells.
Commercial Validation
The pharmaceutical industry has embraced this approach with substantial investments. IDEAYA Biosciences’ clinical collaboration with Amgen represents the first major validation of MAT2A/PRMT5 combinations, bringing together IDEAYA’s IDE397 (MAT2A inhibitor) with Amgen’s AMG 193 (PRMT5 inhibitor) in a joint clinical program.
BeiGene’s recent $150 million acquisition of SYH2039, a MAT2A inhibitor from CSPC Zhongqi Pharmaceutical Technology, demonstrates continued commercial confidence in this approach. BeiGene plans to combine SYH2039 with their own PRMT5 inhibitor BGB-58067, creating another major pharmaceutical partnership focused on this dual-pathway strategy.
These deals reflect more than scientific enthusiasm. They represent calculated business decisions based on preclinical data showing superior efficacy and improved therapeutic windows compared to single-agent approaches. The combination strategy potentially addresses modest response rates that have limited single-agent PRMT5 inhibitors in clinical trials.
Metabolomic Optimization Opportunities
Successful combination development requires sophisticated metabolomic monitoring to optimize dosing, timing, and patient selection. Unlike single-agent therapies, combinations create complex pharmacodynamic interactions that demand pathway-level understanding rather than simple biomarker monitoring.
The key metabolomic parameters include SAM depletion kinetics from MAT2A inhibition, MTA accumulation patterns in MTAP-deleted tumors, and the resulting impact on PRMT5 enzymatic activity. These measurements must be balanced against normal tissue effects to maintain the therapeutic window that makes this approach clinically viable.
Dosing optimization becomes particularly critical because the two drugs operate through different mechanisms with potentially different optimal schedules. MAT2A inhibition may require sustained SAM depletion, while PRMT5 inhibition might benefit from intermittent dosing to minimize toxicity. Metabolomic monitoring can guide these decisions by providing real-time feedback on pathway engagement.
Clinical Development Challenges
The combination approach faces several development challenges that metabolomics can help address. First, patient selection becomes more complex because optimal candidates need both MTAP deletion (for MTA accumulation) and appropriate MAT2A expression levels (for meaningful SAM depletion). Metabolomic profiling can identify patients with the ideal metabolic context for combination therapy.
Second, safety monitoring requires understanding the combined effects on normal cell metabolism. While the therapeutic window should favor cancer cells, comprehensive metabolomic monitoring can detect early signs of normal tissue stress before clinical toxicity becomes apparent.
Third, resistance mechanisms may involve metabolic adaptations that single-biomarker approaches cannot detect. Tumors might compensate for pathway inhibition by upregulating alternative methylation pathways, requiring broad metabolomic surveillance to identify emerging resistance patterns.
Expanding Applications
The MAT2A/PRMT5 combination concept extends beyond MTAP-deleted cancers. Research suggests that some MTAP-intact tumors may still be vulnerable to this approach if they exhibit altered methionine metabolism or elevated MTA levels through other mechanisms. Metabolomic screening could identify these additional patient populations.
Furthermore, the combination strategy might enhance other treatment modalities. Some evidence suggests that methionine restriction or polyamine depletion could create metabolic states that enhance MAT2A/PRMT5 combination efficacy, opening possibilities for triple combinations or dietary interventions.
The approach also has implications for other methyltransferase targets. Similar metabolic dependencies might exist for other SAM-dependent enzymes, potentially expanding the combination concept to additional epigenetic targets beyond PRMT5.
Future Directions
Panome Bio’s PRMT5 Metabolomics Panel provides the comprehensive pathway monitoring needed to optimize these complex combinations. Our assay measures SAM, MTA, and related metabolites alongside downstream products like polyamines and methionine cycle intermediates, providing the complete metabolomic context needed for successful combination development.
The MAT2A/PRMT5 combination approach represents a new paradigm in precision oncology, using metabolic network analysis to identify synthetic lethal interactions that can be therapeutically exploited. Companies that master the metabolomic complexity of these combinations will likely achieve the breakthrough clinical results that single-agent approaches have struggled to deliver.
The convergence of multiple pharmaceutical partnerships around this strategy suggests that MAT2A/PRMT5 combinations may become the standard approach for targeting MTAP-deleted cancers. Success will depend on sophisticated metabolomic monitoring that can unlock the full potential of this promising but complex therapeutic strategy.

