Why Protein-Centric Alzheimer’s Research Has Fallen Short
The pharmaceutical industry’s intensive focus on amyloid-beta and tau pathology in Alzheimer’s disease has yielded limited therapeutic success despite decades of investment and dozens of clinical trials. While these protein aggregates undeniably represent important pathological features of the disease, the repeated failure of compounds targeting amyloid production, aggregation, or clearance suggests that this protein-centric view may be incomplete. Emerging evidence indicates that Alzheimer’s disease involves profound metabolic dysregulation that both precedes and accompanies the formation of classic neuropathological hallmarks, raising the possibility that metabolic dysfunction represents not merely a consequence of neurodegeneration but rather a fundamental driver of disease progression. For drug development programs, this realization necessitates a broader approach to target identification and validation, one that captures the full spectrum of biochemical perturbations underlying cognitive decline rather than focusing exclusively on visible protein deposits.
How Untargeted Metabolomics Expands Alzheimer’s Target Discovery
Unbiased metabolomic profiling provides the comprehensive, systems-level perspective needed to move beyond protein-centric paradigms and identify the full range of metabolic disruptions occurring in Alzheimer’s disease models. By measuring thousands of small molecules simultaneously without presupposing which pathways will prove most relevant, metabolomic approaches can reveal unexpected connections between apparently disparate aspects of disease biology. Recent applications of this technology to ApoE knockout rats have demonstrated the power of comprehensive biochemical phenotyping to uncover lipid metabolism disruptions that fundamentally challenge our understanding of how apolipoprotein E deficiency contributes to Alzheimer’s risk.
ApoE Knockout Models Reveal System-Wide Lipid Dysregulation
When researchers applied Next-Generation Metabolomic® profiling to ApoE knockout rats, they measured over 3,000 distinct metabolites across liver and serum samples (Figure 1), identifying 72 molecules showing statistically significant correlation with ApoE status.
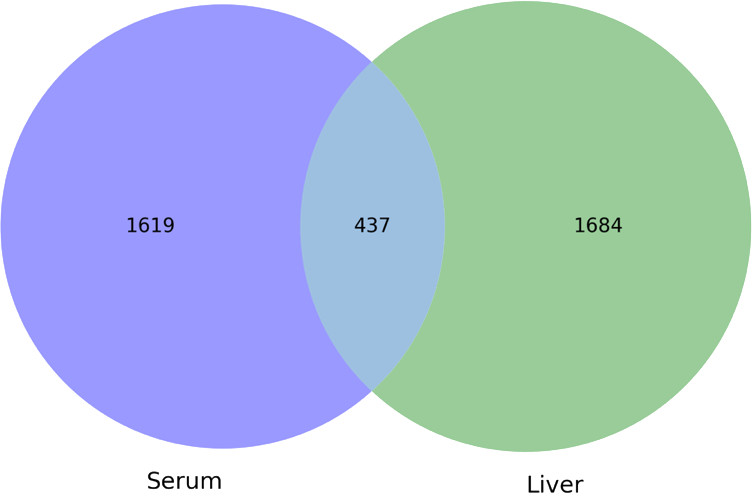
Figure 1: Venn diagram showing unique and overlapping identified metabolites in serum (blue) and liver (green). Only known metabolites from all assays are included.
The pathway enrichment analysis revealed an unexpected finding: rather than perturbations clustering around neurotransmitter metabolism or other traditionally neurodegeneration-associated pathways, the most significantly affected biochemical processes involved lipid metabolism, specifically fatty acylcarnitine (Figure 2) handling and sphingolipid processing. This discovery would have been impossible using hypothesis-driven approaches focused on measuring predetermined sets of molecules related to amyloid or tau pathology, illustrating how unbiased profiling can redirect research attention toward mechanisms that might otherwise remain hidden.
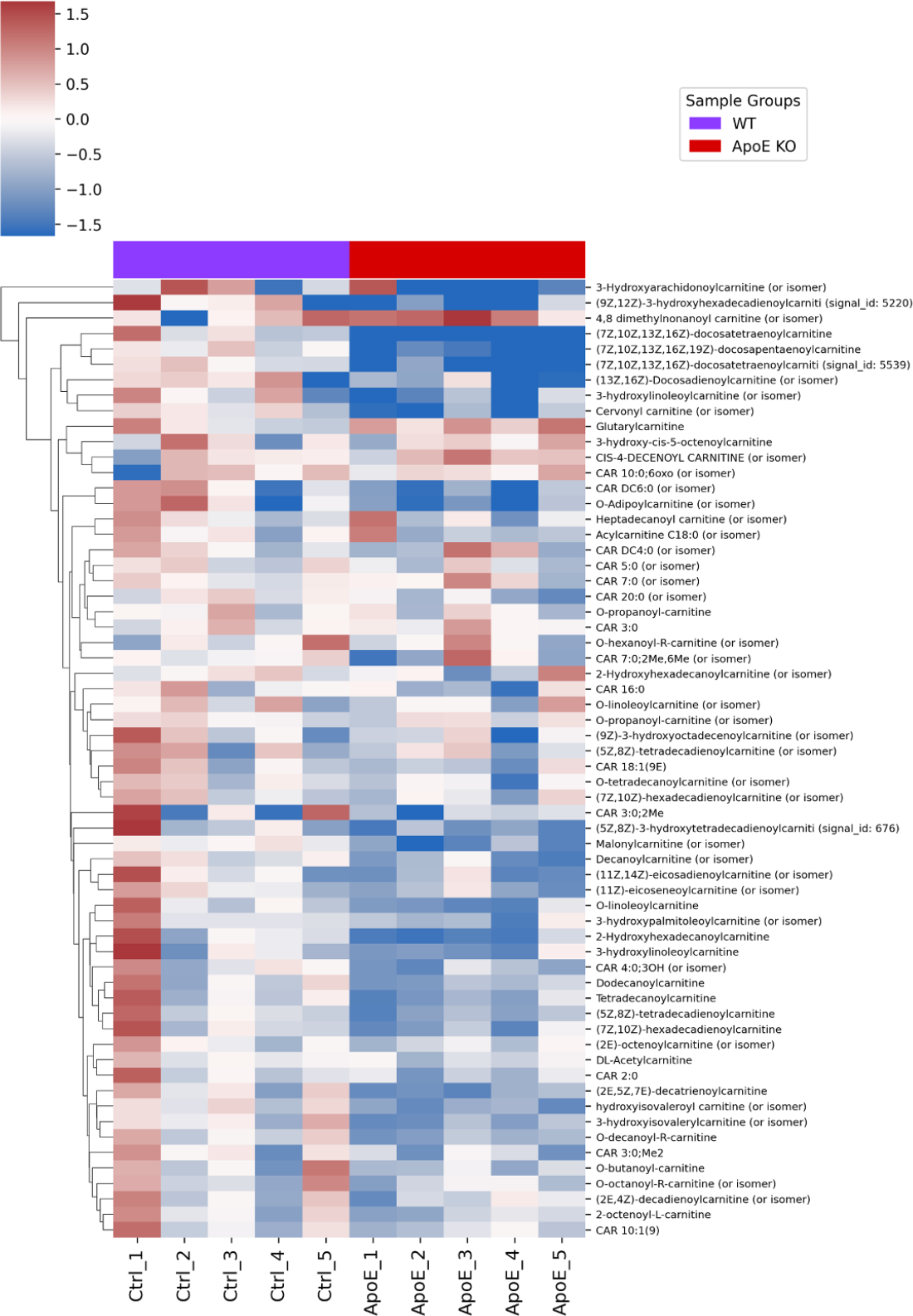
Figure 2: Heatmap of liver fatty acylcarnitines dysregulation showing the log2(fc) relative to the mean level across the samples. Columns are colored according to ApoE status.
The global nature of metabolic disruption observed in these animals extends well beyond the central nervous system, with significant alterations detected in both peripheral circulation and liver tissue. This systemic metabolic phenotype challenges the traditional view of Alzheimer’s disease as primarily a brain disorder and instead suggests a more complex interplay between peripheral metabolic dysfunction and central neurodegenerative processes. For neurodegenerative disease research, this finding opens new avenues for therapeutic intervention targeting peripheral metabolism as a means of supporting brain health, as well as opportunities for peripheral biomarker discovery that could accelerate drug development timelines.
Fatty Acylcarnitine Depletion and Mitochondrial Dysfunction
The identification of widespread lipid metabolism disruptions in ApoE knockout animals aligns with accumulating clinical evidence linking lipid dysregulation to Alzheimer’s disease risk and progression. Epidemiological studies have long documented associations between cardiovascular risk factors (many of which involve lipid metabolism) and later development of dementia, while genetic analyses have implicated multiple lipid metabolism genes beyond APOE in Alzheimer’s susceptibility. The metabolomic characterization of ApoE-deficient rats provides mechanistic insight into these associations by revealing specific lipid species and pathways that become dysregulated in the absence of normal apolipoprotein E function.
The fatty acylcarnitine depletion observed in these animals points to fundamental disruptions in cellular energy metabolism, as these molecules serve essential roles in delivering fatty acids to mitochondria for oxidation. Given the brain’s extraordinary energy demands and its reliance on efficient mitochondrial function, impairments in fatty acid oxidation capacity could have profound consequences for neuronal health and cognitive function. Similarly, the elevation of specific ceramide species suggests dysregulation of sphingolipid-mediated signaling pathways that influence neuronal survival, inflammatory responses, and synaptic function. These lipid metabolism disruptions represent potential therapeutic targets that operate independently of- yet likely interact with – the amyloid and tau pathways that have dominated drug development efforts.
Why Lipid Metabolism Matters for Alzheimer’s Drug Development
The comprehensive metabolomic characterization of preclinical Alzheimer’s models fundamentally changes the landscape of target identification for drug development programs. Rather than limiting therapeutic strategies to modulation of secretase activity, amyloid aggregation, or tau phosphorylation, researchers can now consider interventions targeting the broad array of metabolic pathways shown to be dysregulated in disease-relevant models. For metabolomics contract research organization (CRO) services supporting pharmaceutical development, this shift creates demand for sophisticated biochemical phenotyping that can identify which metabolic perturbations represent viable therapeutic opportunities versus secondary consequences of more fundamental disruptions.
The unbiased nature of metabolomic profiling also provides an opportunity to evaluate existing therapeutic candidates through a new lens. Compounds developed to target amyloid or tau may exert clinically relevant effects through off-target modulation of metabolic pathways, while drugs developed for other indications, diabetes, cardiovascular disease, or metabolic syndrome, might show unexpected benefits in Alzheimer’s models through normalization of lipid metabolism dysregulation. For Alzheimer’s biomarker discovery efforts, the identification of metabolic signatures associated with disease-relevant genetic backgrounds like ApoE deficiency provides candidate biomarkers that could enable patient stratification, disease staging, or monitoring of therapeutic response in clinical trials.
Integrating Lipidomics into Alzheimer’s Drug Development
The revelation that lipid metabolism disruptions represent a central feature of Alzheimer’s disease models argues for systematic integration of metabolomic profiling throughout the drug development pipeline. At the earliest stages of target identification, comprehensive metabolic phenotyping of disease models can reveal unexpected therapeutic opportunities that would not emerge from protein-centric screening approaches. During lead optimization, lipidomics services can assess whether candidate compounds successfully normalize disease-relevant metabolic signatures, providing early evidence of pharmacodynamic activity before investing in lengthy efficacy studies. In translational research, metabolomic biomarkers identified in preclinical models can be evaluated in clinical samples, potentially enabling more efficient patient selection and response monitoring strategies.
The systems biology perspective enabled by metabolomic profiling also facilitates identification of combination therapy opportunities. Given that lipid metabolism disruptions intersect with mitochondrial dysfunction, inflammatory signaling, vascular pathology, and ultimately protein aggregation, metabolic phenotyping can guide rational combination strategies where lipid-targeting approaches complement interventions aimed at other aspects of disease biology. For research teams seeking to move beyond failed protein-centric paradigms, Panome Bio’s Next-Generation Metabolomics services provide the comprehensive biochemical characterization necessary to identify promising metabolic targets and develop therapeutics that address the full spectrum of Alzheimer’s disease pathology.