Alzheimer’s as a System-Level Disorder via Multi-Omics
Alzheimer’s disease is not driven by a single pathway, molecule or mutation. Instead, it reflects a system-level breakdown across metabolism, protein regulation and cellular signaling. Yet most studies still analyze these layers in isolation.
That’s the gap Multi-Omics is designed to close.
Multi-Omics integrates complementary biological layers, such as untargeted metabolomics and proteomics workflow, to reveal how molecular systems function together in health and disease. By combining Next-Generation Metabolomics® and Discovery Proteomics, Panome Bio captures how those changes are connected across biology.
From Molecules to Mechanisms in Alzheimer’s Disease
Panome Bio applied its integrated Multi-Omics workflow to serum samples from 15 Alzheimer’s disease patients and 15 healthy, age- and sex-matched controls.
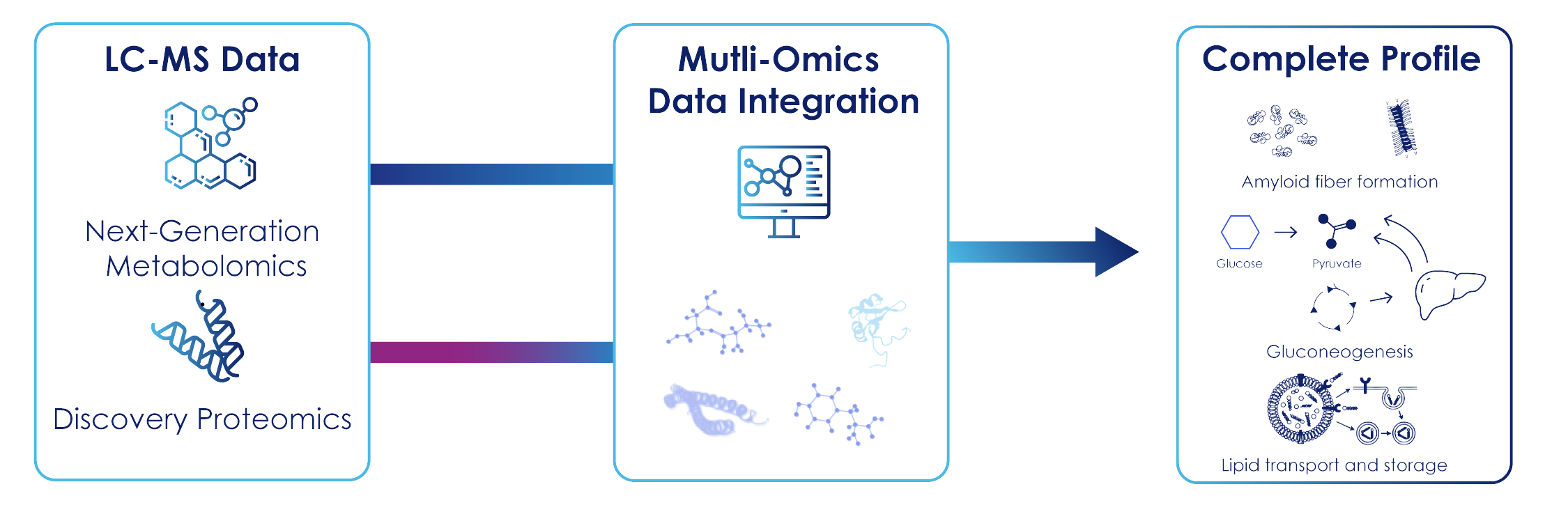
Figure 1: Integrated Multi-Omics Workflow in Alzheimer’s Disease: Integrated Multi-Omics workflow combining Next-Generation Metabolomics and Discovery Proteomics into a unified biological profile. Advanced computational integration maps Alzheimer’s disease-associated signatures, including amyloid fiber formation, dysregulated glucose metabolism and lipid transport alterations.
Alzheimer’s Disease Metabolomics Reveals Energy Collapse
- 6,115 metabolites profiled
- 1,062 significantly altered
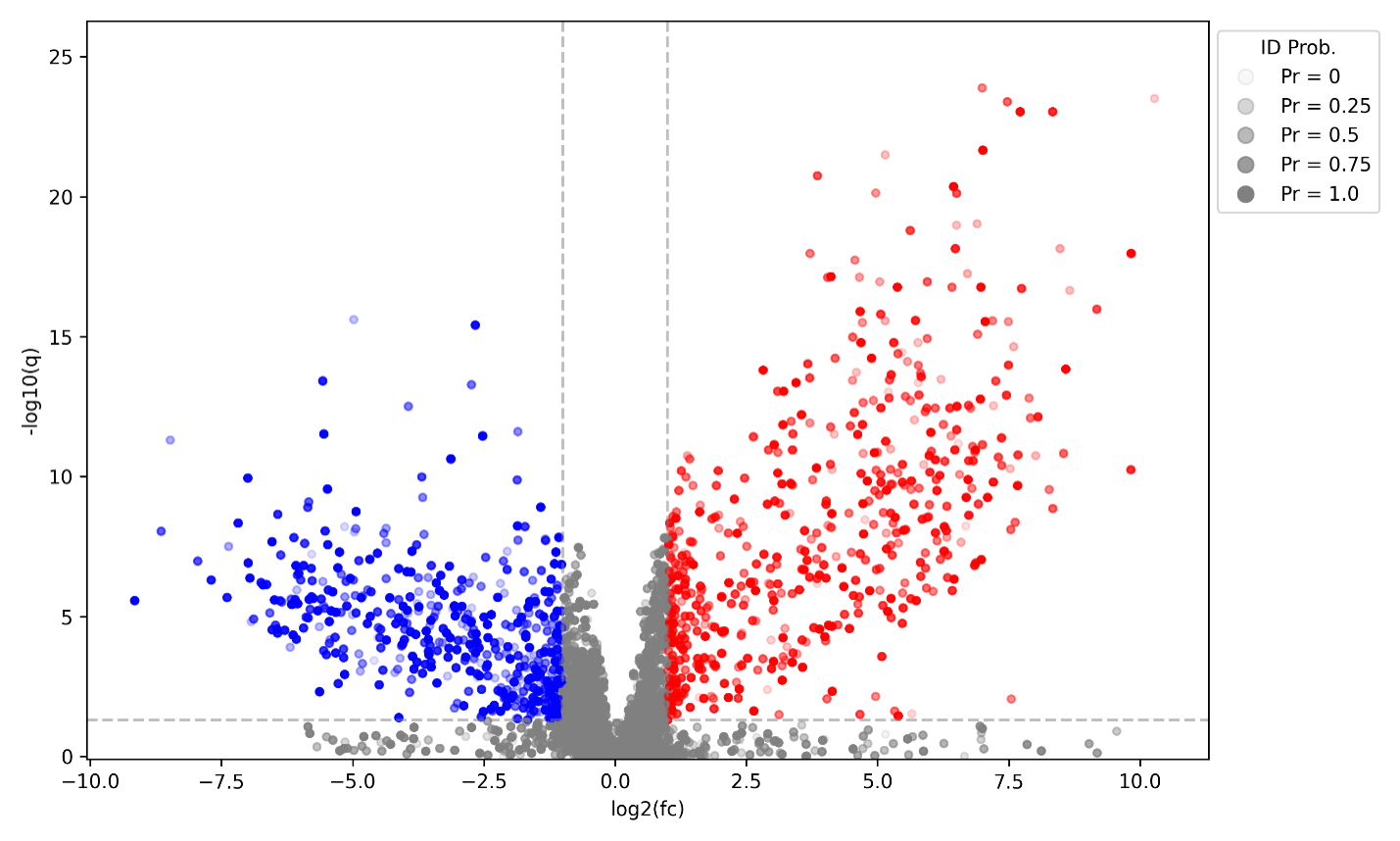
Figure 2: Volcano Plot of Differential Metabolite Expression in Alzheimer’s Disease: Each point represents a metabolite plotted by log2 fold change and statistical significance, highlighting significantly upregulated (red) and downregulated (blue) features. Dot shading reflects identification confidence.
Key Findings
- Accumulation of mitochondrial stress lipids (cardiolipins, gl ycerolipids)
- Depletion of energy metabolites (lactate, malate, glycerate)
- Loss of bile acids and lipid-soluble vitamins
Why This Matters
These changes reflect a system under severe metabolic stress:
- Impaired energy production
- Disrupted membrane integrity
- Breakdown of metabolic homeostasis
Disruptions in energy metabolism and mitochondrial function are well established in Alzheimer’s disease and are considered central to disease progression.
Alzheimer’s Disease Proteomics Identifies Molecular Drivers
- 2,362 protein groups quantified
- 552 significantly altered
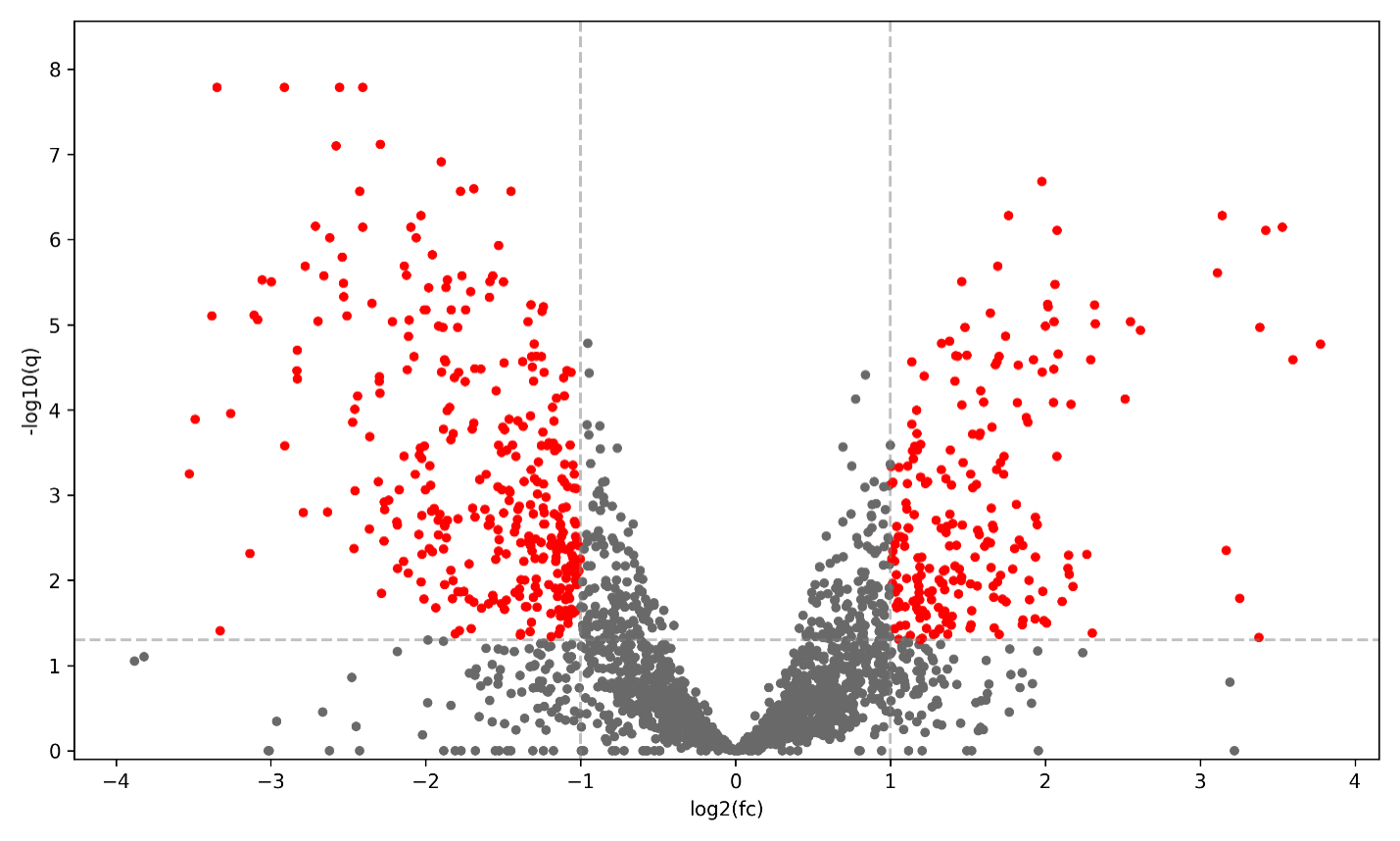
Figure 3: Protein Volcano Plot in Alzheimer’s Disease: Volcano plot of differential protein expression in Alzheimer’s disease. Each point represents a protein plotted by log2 fold change and statistical significance between disease and control samples. Proteins meeting significance thresholds are shown in red, highlighting key proteomic alterations associated with disease pathology.
Key Findings
- Downregulation of ApoE and ApoA1
- Upregulation of inflammatory and vascular markers (e.g., fibrinogen-related proteins)
ApoE plays a central role in:
- Lipid transport
- Amyloid clearance
- Neuronal repair
Why This Matters
Reduced ApoE levels provide a mechanistic link to:
- Lipid dysregulation
- Plaque accumulation
- Progressive neurodegeneration
These findings align with prior studies linking ApoE-mediated lipid transport dysfunction to Alzheimer’s disease pathology.
Why Integrating Omics Changes the Scale of Discovery
Studying metabolomics or proteomics alone provides only partial insight. Integrating them transforms the scale of discovery, into a unified biological profile. Advanced computational analysis maps disease-associated signatures in Alzheimer’s disease, including amyloid fiber formation, dysregulated glucose metabolism and altered lipid transport and storage.
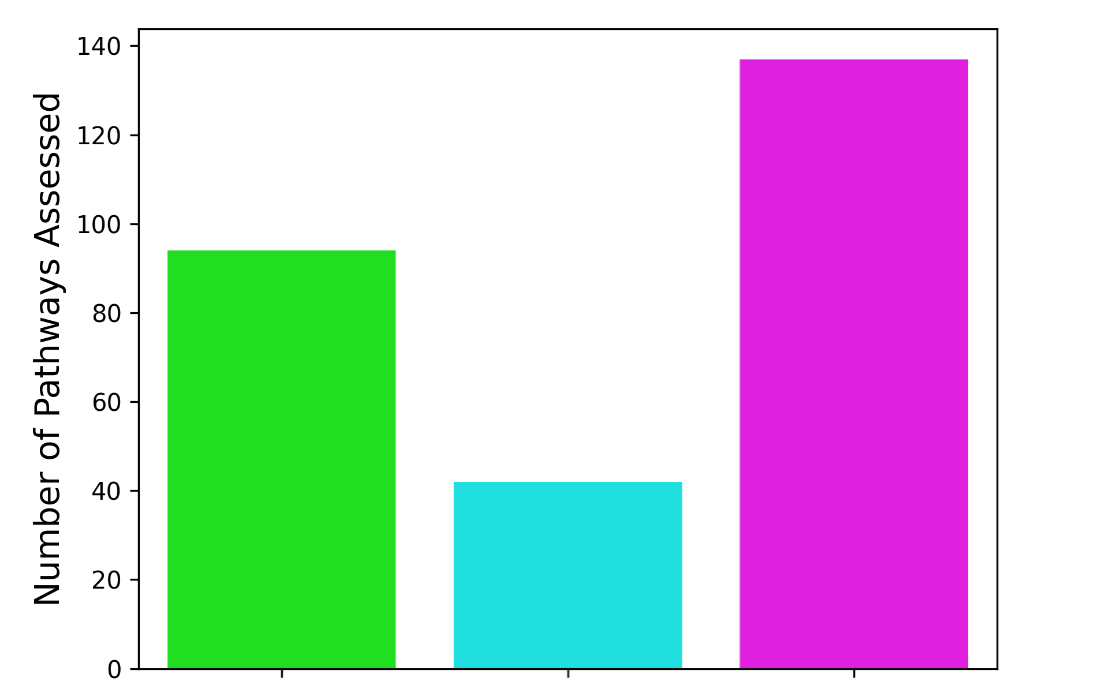
Figure 4: Number of pathways available from individual and integrated omics in Alzheimer’s Disease: Comparison of biochemical pathway coverage using individual and integrated molecular profiles in Alzheimer’s disease. The analysis shows the number of detectable pathways using proteomics alone, metabolomics alone, and the combined multi-omics dataset, demonstrating that integration significantly expands biological pathway discovery and interpretability.
Pathway-Level Changes
- 40+ additional pathways identified through integration
- 25 significantly enriched biochemical pathways
Key Pathways Include
- Amyloid fiber formation
- Glucose metabolism and gluconeogenesis
- Lipid transport and storage
These pathways highlight the interplay between amyloid biology, oxidative stress, and metabolic dysfunction in Alzheimer’s disease.
Network Analysis Reveals Biology as a System
When proteins and metabolites are combined into a unified network:
- 102 metabolites + 83 proteins are connected
- Distinct biological modules emerge
Key Network Modules
- Amyloid-related pathways
- Energy metabolism (gluconeogenesis)
- Lipid regulation systems
This shifts Multi-Omics from measurement to mechanism showing how systems interact.
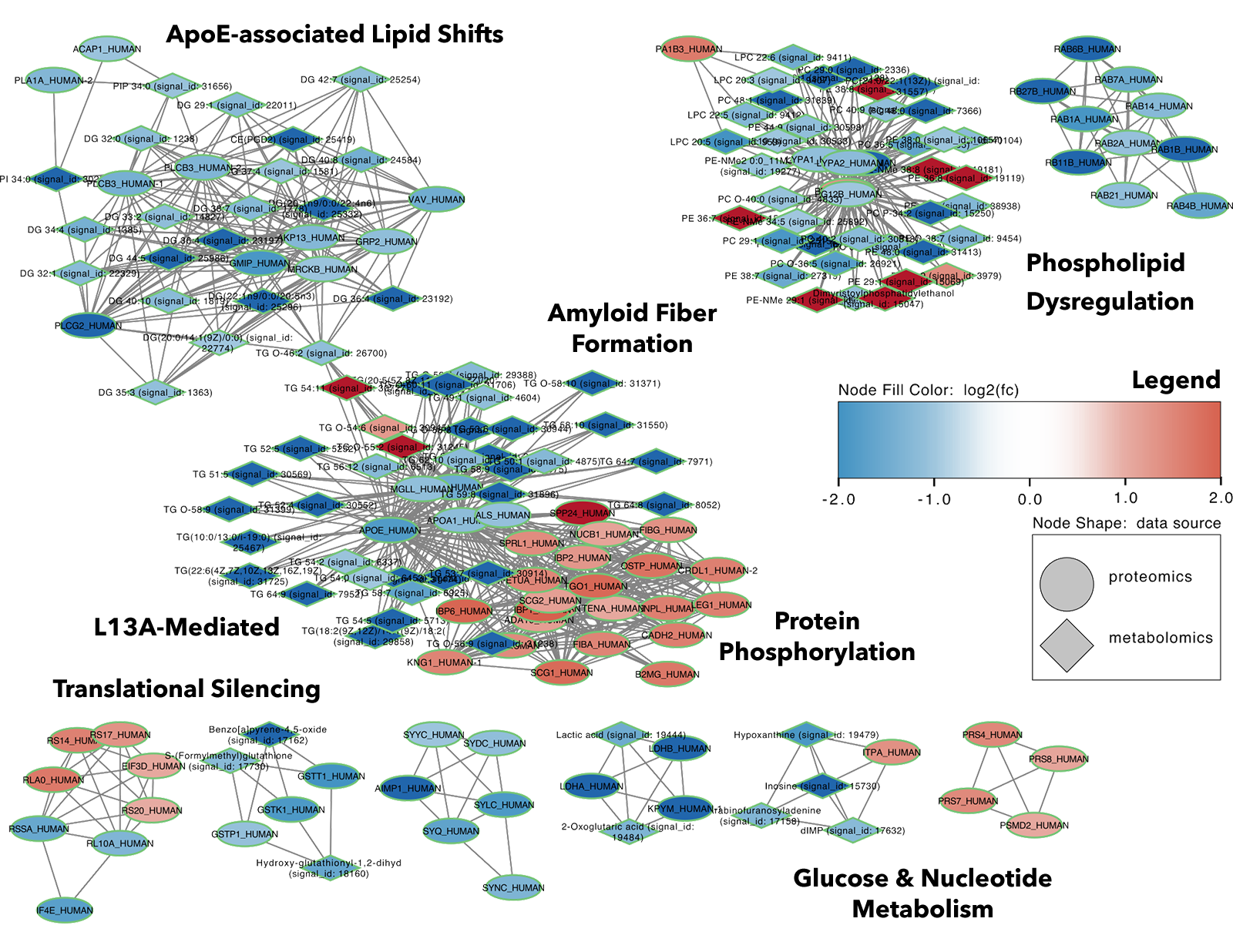
Figure 5: Integrated Multi-Omics Network Analysis in Alzheimer’s Disease: Integrated network analysis combining metabolites and proteins in Alzheimer’s disease. Nodes represent metabolites and proteins, colored by log2 fold change between disease and control samples. The resulting network reveals system-level disease organization, highlighting coordinated molecular interactions across metabolic and proteomic layers.
A Global Molecular Signature of Alzheimer’s Disease
- 11,150 total analytes measured
- 1,618 significantly altered between Alzheimer’s and healthy samples
- 25 enriched biochemical pathways identified
- Clear separation between disease and control groups
Instead of isolated molecular changes, Multi-Omics reveals coordinated biological programs moving together.
Alzheimer’s disease is not subtle, it is a coordinated system-wide disruption.
What’s Driving Alzheimer’s at the Molecular Level?
The integrated data reveals a consistent biological theme: a system under metabolic and lipid stress, compounded by vascular dysfunction.
Key Mechanisms Identified
- Disrupted lipid transport (ApoE, ApoA1 reduction)
- Mitochondrial stress (cardiolipin accumulation)
- Energy depletion (loss of lactate and malate)
- Vascular dysfunction (increased fibrinogen-related proteins)
How These Processes Connect
- Reduced ApoE impairs lipid transport and amyloid clearance
- Lipid imbalance increases mitochondrial stress
- Energy production declines
- Vascular dysfunction accelerates disease progression
Together, these interactions form a reinforcing cycle of neurodegeneration.
The Panome Bio Approach
Panome Bio enables system-level discovery through:
- Next-Generation Metabolomics : Broad coverage across lipid, polar, and intermediate chemistries
- Discovery Proteomics: Deep, system-wide protein quantification
- Integrated Multi-Omics Analysis : Connects molecules into pathways, networks, and mechanisms
Each layer is powerful on its own. Together, they create a unified view of disease biology.
Where Multi-Omics Makes an Impact
While this study focuses on Alzheimer’s disease, the same integrated approach is broadly applicable across biology and medicine:
- Neurodegeneration
- Cancer biology
- Drug response variability
- Animal Model Research
- Metabolic disease
- Systems biology research
- Cultured Meat Production
Multi-Omics is not disease-specific, it is system-specific.
Ready to See the Full Picture?
Disease does not operate in silos.
Neither should your data.
With integrated Multi-Omics, Panome Bio delivers the depth, clarity and connectivity needed to move beyond observation and into true biological understanding.