In the fight against cancer, single-agent treatments often hit a wall as tumors adapt and develop resistance. This is especially true for aggressive cancers like non-small-cell lung cancer (NSCLC) with KRAS and LKB1 mutations, a hard-to-treat subtype. These challenges are fueling a critical shift in oncology towards combination therapies that target cancer from multiple angles.
Panome Bio’s recent collaboration with TD2 Oncology, a leader in translational drug development, highlights this paradigm shift. Using Panome Bio’s Next-Generation Metabolomics® platform, combination of two targeted drugs creating a powerful, synergistic effect was explored in a preclinical model of KRAS/LKB1-mutant NSCLS.
The Challenge: Overcoming Resistance in KRAS-Mutant NSCLC
KRAS mutations are found in a significant portion of cancers, especially in NSCLC often associated with poor prognosis. When combined with LKB1 loss, tumors become even more resistant and difficult to treat. One promising strategy is to combine MEK inhibitors, which target a key signaling pathway, with histone deacetylase (HDAC) inhibitors, which reserve epigenetic silencing.
By promoting histone acetylation and relaxing chromatin structure, HDAC inhibitors enable transcription of genes that drive cell cycle arrest, apoptosis, and/or immune modulation, leaving tumors more vulnerable.
The following study focuses on a combination of Trametinib (a MEK inhibitor) and GB-3103 (a Class I/IIb HDAC inhibitor). Using an A549 NSCLC xenograft mouse model, Panome Bio compares GB-3103 alone, Trametinib (a MEK inhibitor) alone, and the combination, via untargeted metabolomics.
MEK and HDAC Inhibitor Combination in NSCLC
The results are clear: while both drugs have some effect on tumor growth individually, the combination outperforms either drug alone. The real question is why and how?
This is where metabolomics comes in. By analyzing the metabolic changes in the tumor, Panome Bio uncovers precise mechanisms behind this potent synergy.
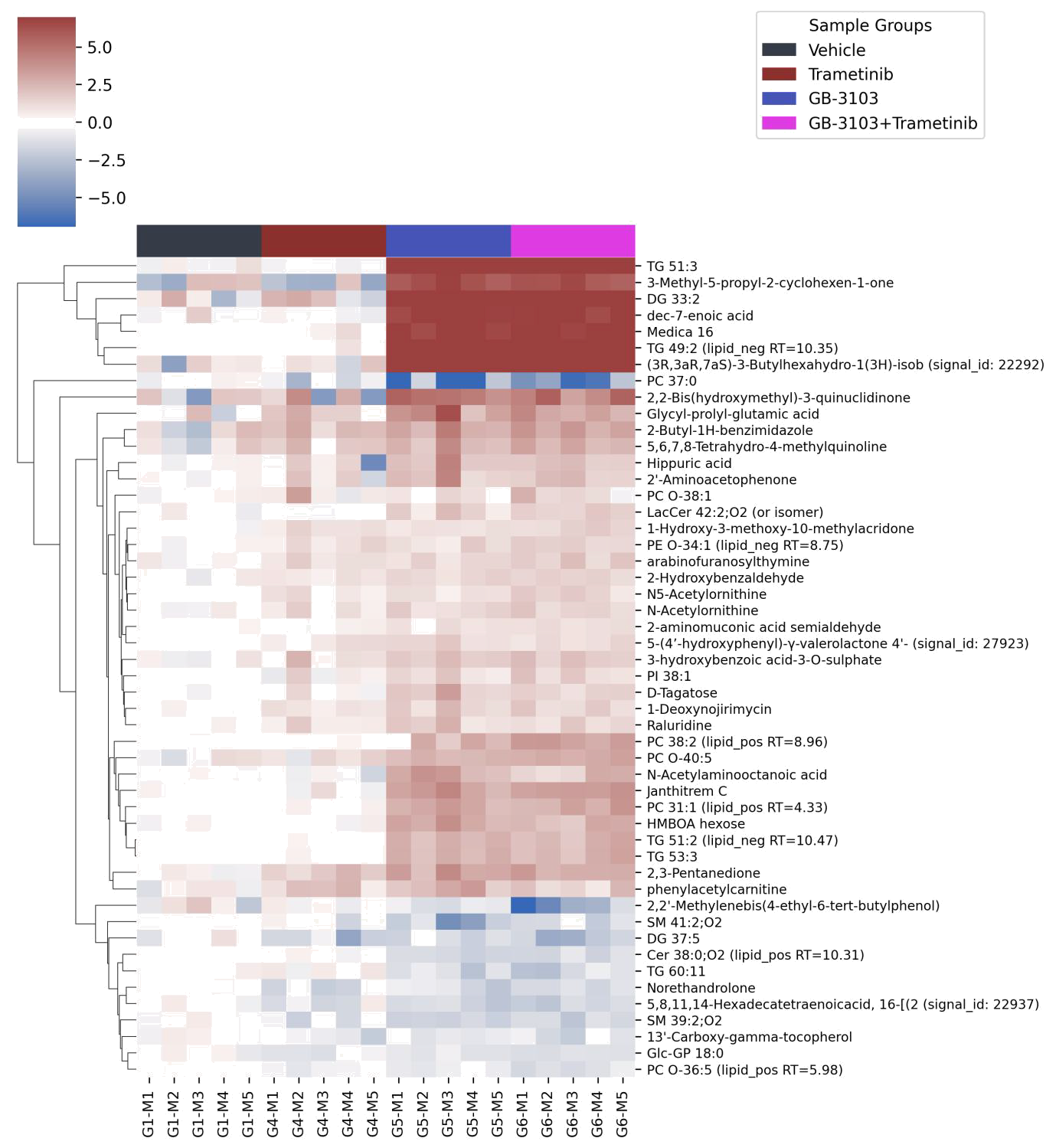
Figure 1: Heatmap of top 50 significantly altered metabolites.
Across 3,942 metabolites identified (2.300 lipids and 1,642 polar compounds), the combination treatment drove a much broader and more profound metabolic disruption than either monotherapy.
- 19 significantly altered metabolic pathways.
- Enriched: Simple Glucose (Glc) series, fatty acid estolides.
- Depleted: Diacylglycerophosphoinositols, gangliosides.
- Depleted metabolites: Lipids like PC O-36:5 and triglycerides (TG 13:0/13:0/13:0) indicated impaired membrane synthesis and energy storage.
- Amino acid shifts: Decreased cystathionine levels suggested altered amino acid metabolism and oxidative imbalance.
- Elevated metabolites: Acetyl lysine and cystathionine (Figure 6) reflected metabolic and oxidative stress, suggesting tumor cells were burdened beyond compensatory capacity.
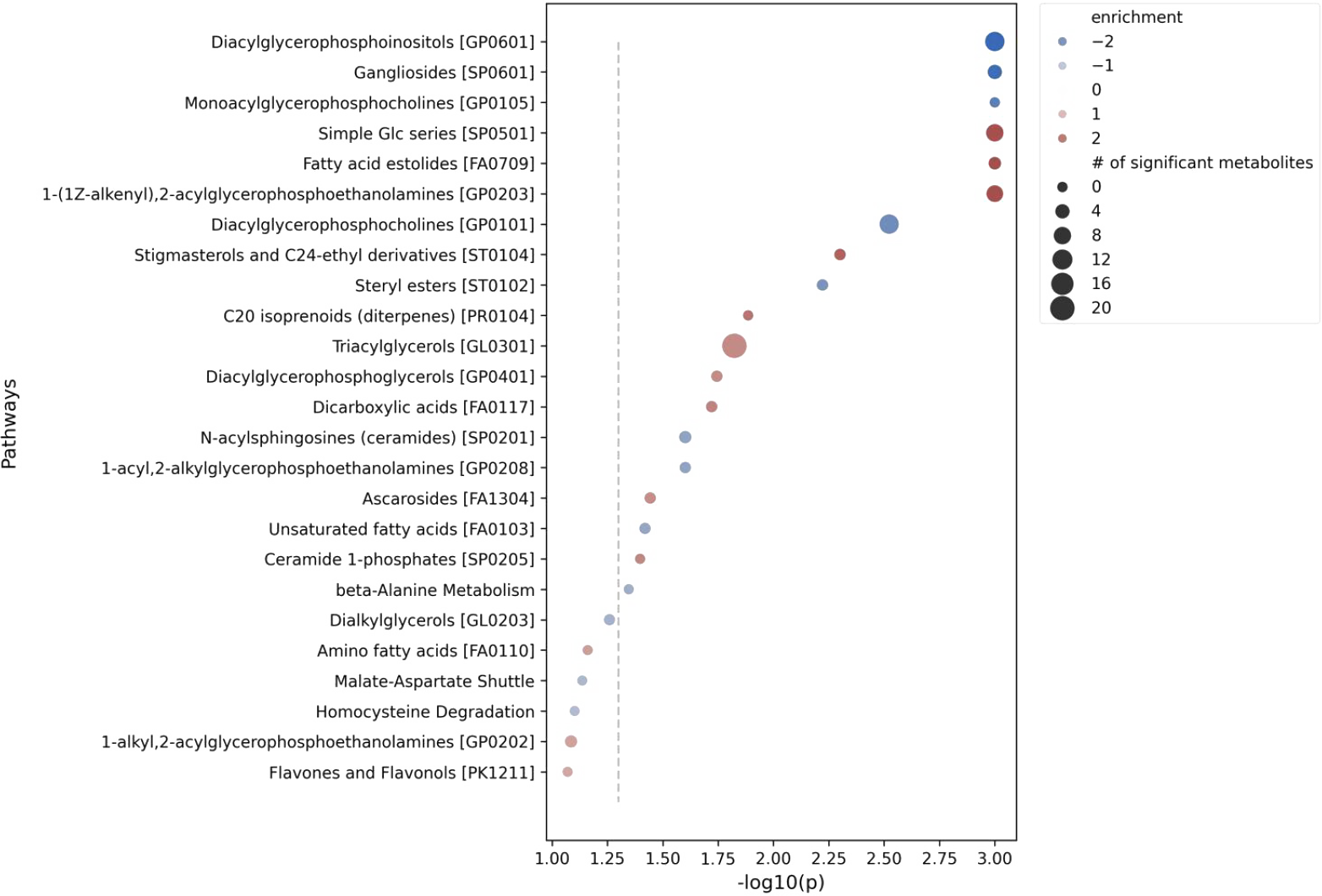
Figure 2: Pathway analysis of significant metabolites.
The metabolic shifts weren’t only a simple addition of the two drugs’ effects. Instead, they revealed a true synergistic disruption of the tumor’s biology. The combination didn’t just block one pathway, it created a state of deep, multifaceted metabolic stress, cutting off survival mechanisms. Untargeted metabolomics not only revealed why this combination works but also highlighted biomarkers and pathways for future therapeutic strategies.
What Does This Mean for Cancer Therapy?
Through this collaboration with TD2 Oncology, Panome Bio provided insights from this study into how synergistic drug combination works. By creating a metabolic blueprint of the combination’s effect, researchers can now:
- Identify predictive biomarkers for patient stratification: These metabolic markers can help researchers identify which patients are most likely to respond to combination therapy.
- Optimize combination strategies: The data can guide the development of even more effective drug cocktails by targeting specific metabolic vulnerabilities.
- Explore new research frontiers: These findings suggest that HDAC inhibitors may have broader potential when combined with other targeted therapies to induce metabolic distress across different cancer types.
Moving forward, a multi-omics approach will be key. By integrating data from metabolomics with proteomics and phosphoproteomics, researchers can create a complete map, for example on how HDAC and MEK inhibition converge on critical cellular processes. This holistic view will provide even deeper insights, leading to more precise, effective, and personalized cancer treatments.
Panome Bio provides the tools and expertise to reveal the hidden metabolic stories in your data, helping you give your research direction and translate complexity into clarity.
Why Partner with Panome Bio?
At Panome Bio, we specialize in helping our partners get the most out of their data. Our state-of-the-art platform offers:
- True Untargeted Discovery: We identify both known and novel metabolites without limitations, using a database over 175,000 compounds.
- AI-Powered Precision: Our proprietary algorithms cut through noise to deliver high-confidence, biologically relevant results.
- Multi-Omics Integration: We combine metabolomics, proteomics, and transcriptomics to offer deep pathway coverage and powerful, integrated information.