The Lipid Connection in Neurodegeneration
While much of Alzheimer’s disease research has historically focused on protein aggregation (amyloid-beta plaques and tau tangles), a growing body of evidence points to fundamental disruptions in lipid metabolism as both driver and consequence of neurodegenerative processes. Among the various lipid classes implicated in Alzheimer’s pathology, sphingolipids have emerged as particularly significant players, with ceramides serving as critical signaling molecules that influence neuronal survival, inflammatory responses, and the very protein aggregation pathways that define the disease. The challenge for pharmaceutical researchers has been determining whether sphingolipid dysregulation represents a viable therapeutic target or simply an epiphenomenon of broader metabolic collapse, and whether preclinical models faithfully recapitulate the specific alterations in ceramide metabolism observed in human patients.
Recent advances in Alzheimer’s disease metabolomics and lipidomics services have enabled unprecedented resolution in mapping lipid perturbations across disease models and clinical samples. When applied to the characterization of ApoE knockout rats, comprehensive metabolomic profiling has revealed striking elevations in ceramide species alongside broader disruptions in sphingolipid metabolism pathways. These findings carry profound implications for both our understanding of how apolipoprotein E deficiency contributes to Alzheimer’s risk and for the validation of preclinical models used in neurodegenerative disease research.
Figure 1: Study design and analysis workflow.
Ceramide Elevation: A Metabolic Signature with Mechanistic Consequences
Pathway enrichment analysis of metabolomic data from ApoE knockout rats revealed significant dysregulation of sphingolipid metabolism in both serum and liver samples, with eight distinct ceramide species showing statistically significant elevation compared to wild-type controls. This metabolic signature directly parallels extensive clinical observations documenting increased ceramide levels in the brains and peripheral circulation of Alzheimer’s disease patients, where elevated basal ceramide concentrations have been associated with increased disease risk and more rapid cognitive decline. The spontaneous development of this sphingolipid phenotype in ApoE-deficient animals – without additional genetic manipulation or dietary intervention – suggests that the model naturally engages disease-relevant lipid metabolism pathways.
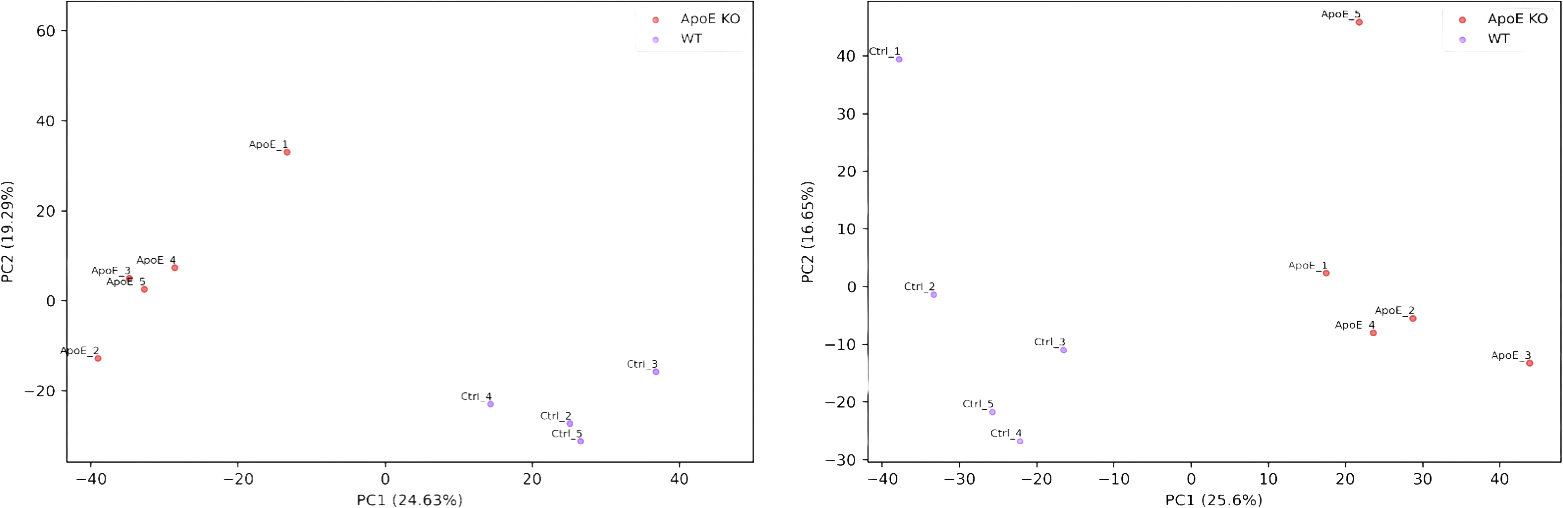
Figure 2: Unsupervised principal components analysis (PCA) visualizes the global metabolite profiles of liver (left) and serum (right) samples. Together it highlights clear trends and separation between groups, reflecting distinct metabolite concentration patterns in both tissues.
The mechanistic implications of ceramide elevation extend well beyond simple biomarker correlation. Ceramides influence the processing of amyloid precursor protein through effects on β-secretase and γ-secretase activity, potentially increasing the production of pathogenic amyloid-beta peptides. Additionally, these bioactive lipids activate stress-activated protein kinases and promote mitochondrial dysfunction, creating a cellular environment conducive to neuronal apoptosis. Emerging evidence also suggests ceramide involvement in tau hyperphosphorylation, linking sphingolipid dysregulation to both major pathological hallmarks of Alzheimer’s disease. For drug development programs, this mechanistic connectivity positions ceramide metabolism as an attractive intervention point where single compounds might influence multiple disease pathways simultaneously.
Sphingolipid Pathway Complexity: From Synthesis to Catabolism
Metabolomic analysis revealed dysregulation across multiple nodes of sphingolipid metabolism, suggesting systemic rather than focal pathway disruption. The sphingomyelinase pathway showed significant alterations, with specific sphingomyelin species elevated in ApoE knockout animals, while the downstream catabolic pathway demonstrated increased phosphoethanolamine levels. This pattern suggests enhanced flux through ceramide-generating pathways, potentially driven by increased sphingomyelin hydrolysis or accelerated de novo synthesis. The complexity of these interconnected pathways underscores the value of comprehensive metabolomic approaches over targeted measurements of individual lipid species, as perturbations at any point in the network can have cascading effects throughout the system.
For researchers conducting Alzheimer’s biomarker discovery or developing therapeutics targeting lipid metabolism, this pathway-level view provides critical context for experimental design and data interpretation. Interventions aimed at reducing ceramide levels might be achieved through multiple approaches: inhibiting de novo synthesis, blocking sphingomyelin hydrolysis, or accelerating ceramide clearance through the catabolic pathway. The ApoE knockout model’s recapitulation of human sphingolipid dysregulation patterns makes it a relevant system for evaluating which intervention strategy most effectively normalizes the metabolic signature while improving cognitive and neuropathological outcomes.
Peripheral Metabolic Signatures as Windows into Central Pathology
One particularly valuable aspect of the sphingolipid dysregulation observed in ApoE knockout rats is its detectability in peripheral tissues and circulation. The significant enrichment of ceramides and sphingolipid pathway metabolites in serum samples provides opportunities for longitudinal monitoring throughout preclinical studies without requiring invasive central nervous system sampling. This peripheral accessibility addresses a persistent challenge in neurodegenerative disease research: obtaining objective, quantitative readouts of disease state and therapeutic response in a manner compatible with repeated measurements and high-throughput screening.
The correlation between peripheral sphingolipid alterations and central pathology, documented in both human Alzheimer’s patients and now validated in the ApoE knockout model, suggests that serum ceramide measurements could serve as pharmacodynamic biomarkers during preclinical development. For lipid profiling studies in drug development, this enables early assessment of whether candidate therapeutics successfully engage their intended targets and normalize disease-relevant metabolic pathways. The metabolomics CRO approach of comprehensive profiling also allows detection of unintended off-target effects on lipid metabolism, potentially identifying liabilities before advancing compounds into costly efficacy studies.
Implications for Therapeutic Development
The metabolomic validation of sphingolipid dysregulation in ApoE knockout rats strengthens the case for therapeutic strategies targeting ceramide metabolism in Alzheimer’s disease. The convergence between the model’s metabolic phenotype and documented human disease signatures reduces the translational risk inherent in preclinical drug development, providing confidence that efficacy signals observed in ApoE-deficient animals are more likely to reflect therapeutically relevant mechanisms. For research teams evaluating sphingolipid-modulating compounds, this model offers a validated platform for proof-of-concept studies that integrates metabolic biomarker readouts with traditional cognitive and histopathological endpoints.
The comprehensive nature of metabolomic profiling also enables identification of combination therapy opportunities. Given that sphingolipid dysregulation intersects with mitochondrial dysfunction, inflammatory signaling, and protein aggregation pathways, metabolic phenotyping can guide rational combination strategies where ceramide-lowering approaches are paired with complementary interventions targeting other aspects of disease biology. For preclinical Alzheimer’s models, Panome Bio’s Next Generation Metabolomics services provide the systems-level biochemical characterization necessary to identify such synergistic opportunities and optimize therapeutic strategies for maximum translational potential.

